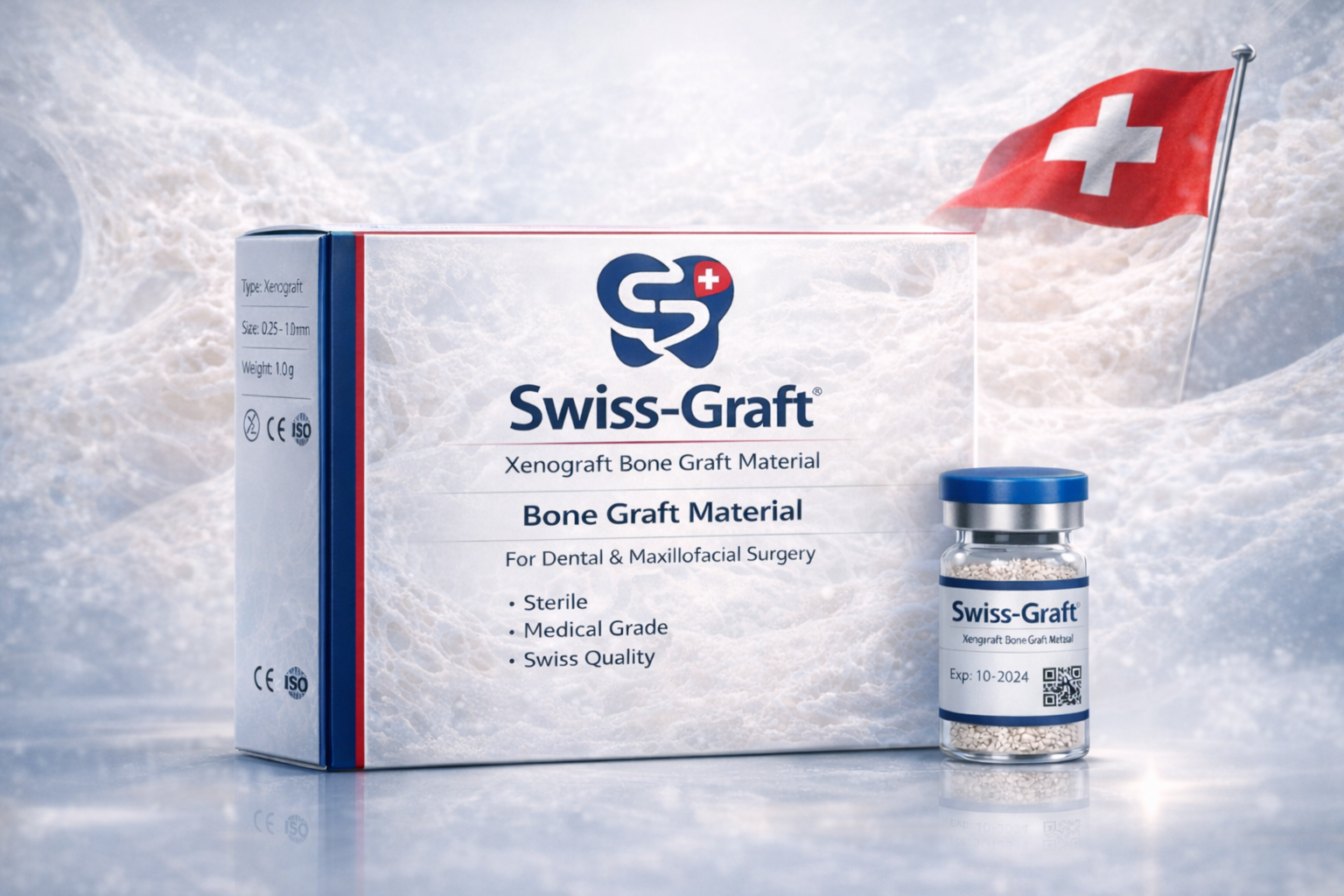
Product Name: Swiss-Graft® Xenograft Bone Graft
Source: Premium bovine cortical bone
Particle Size: 0.25 – 1 mm (250 – 1000 µm)
Country of Origin: Switzerland 🇨🇭
Shelf Life: 3 years
Certifications: CE Class II – ISO 13779 – ISO 5833 – ISO 13485 – FDA Registered
🟦 Available Sizes:
All sizes are pre-sterilized and packaged in double-barrier systems.
Description:
Swiss-Graft® Xenograft represents the pinnacle of bone regeneration technology. Derived from premium bovine cortical bone, it undergoes a sophisticated multi-stage processing protocol that removes all organic components while preserving the critical mineral architecture. This unique processing maintains the natural porosity and bioactivity of the bone structure, promoting optimal cellular integration and new bone formation.
🔬 Technical Specifications:
- Particle Size: 250 – 1000 µm
- Porosity: 75 – 85%
- Density: 2.0 – 2.3 g/cm³
- Bioactivity: Grade A
- Compressive Strength: 15 – 25 MPa
- Resorption Rate: 12 – 18 months
🔹 Clinical Applications:
- Dental implant site preparation and alveolar bone deficiencies
- Ridge augmentation (horizontal and vertical)
- Extraction socket preservation
- Guided Bone Regeneration (GBR)
- Maxillary sinus lift procedures
- Periodontal bone defect repair
- Maxillofacial reconstruction and trauma sites
🔹 Key Benefits:
- Pre-hydrated granules with excellent moldability – no fixation agents needed
- Exceptional biocompatibility with minimal inflammatory response
- Predictable resorption within 12 – 18 months with simultaneous new bone formation
- Multi-stage sterilization (ISO 11135) – minimizes infection risk
- 95% osseointegration rate proven in clinical studies
- Swiss precision engineering – consistent batch-to-batch performance